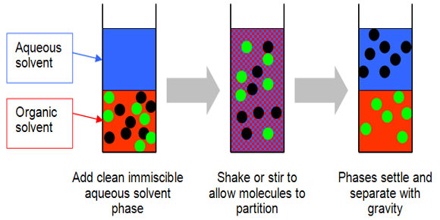
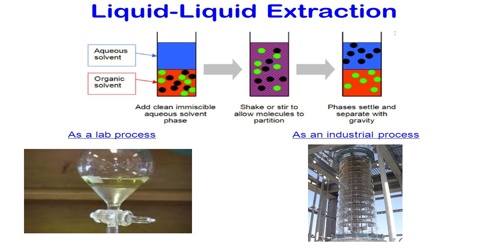
Liquid-liquid extraction LLE is one of the most commonly used techniques for sample preparation which allows the selective concentration of target analytes in the aqueous samples. One liquid phase is the feed consisting of a solute and a carrier.

Chem117 04 Liquid Liquid Extraction Fundamentals Youtube
Solids liquids and gases The particle theory is used to explain the properties of solids liquids and gases.
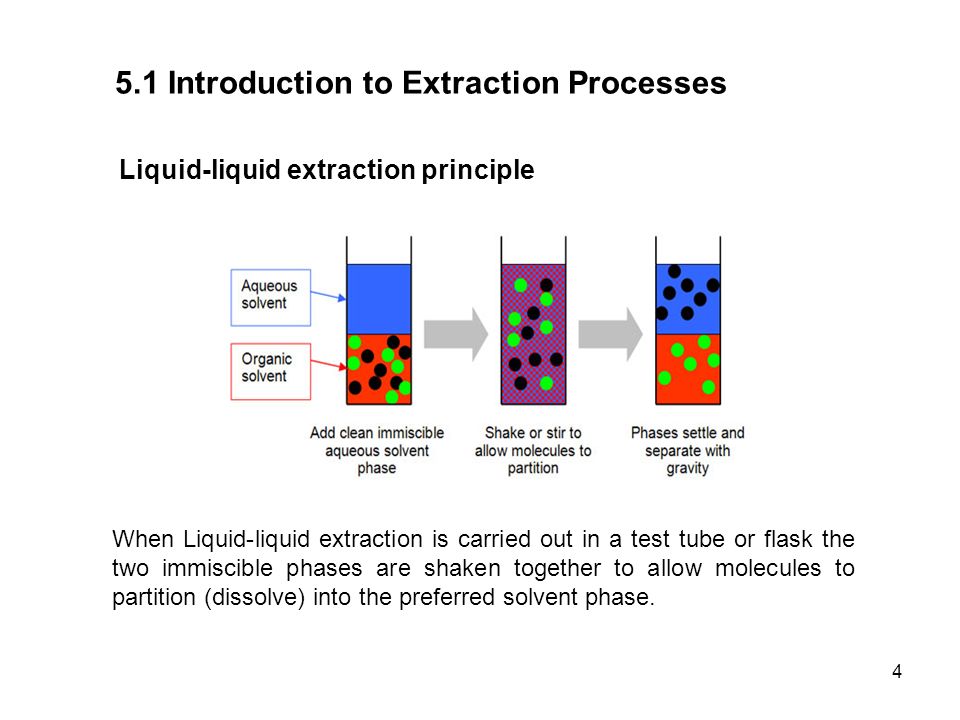
. The extraction technique can be used to purify compounds or to separate mixtures of compounds such as when isolating a product from a reaction mixture known as an extractive work-up. The other phase is the solvent. Number of Stages 630 Example.
Usually we are interested to separate only one component from a feed stream. With equilibrium and non-equilibrium molecular dynamics simulations we illustrate the kinetics of solute crossing the interface and in particular the structure of the interface at which two stagnant liquid films adhere. B has to be extracted into another liquid C.
Liquid-liquid or solvent extraction is a countercurrent separation process for isolating the constituents of a liquid mixture. In this procedure the organic product is isolated from inorganic substances. Mixing and Operating Points 930 Hunter Nash Method 2.
This item is available to borrow from 1 library branch. 1000 kghr of a feed containing 30 wt acetone. The organic solvent used for extraction must meet a few criteria.
Slower than the reaction rateThe liquid-liquid extraction is a mass transfer process between two phases. 4 LIQUID-LIQUID EXTRACTION PROCESS 41 General Liquid-Liquid Extraction is the process of extracting a Solute from a Feed by use of a Solvent to produce an Extract and a Raffinate. This is related to a mercury electrode where a metal can be reduced the metal will often then dissolve in the mercury to form an amalgam that modifies its electrochemistry greatly.
The extraction is understood to be a transfer of the solute from the feed to the solvent. The Feed stream may consist of any number of components. Liquid-liquid extraction also known as partitioning is a separation process consisting of the transfer of a solute from one solvent to another the two solvents being immiscible or partially miscible with each other.
The first choice in designing any liquid extraction is the choice of extracting solvent and this choice is driven by the relative hydrophobicity of the analyte molecule which is reflected by its LogP D value. In a system consisting of a molten metal in contact with molten salts metals can be extracted from one phase to the other. In its simplest form this involves the extraction of a solute from a binary solution by bringing it into contact with a second immiscible solvent in which the solute is.
During extraction the solute travels from liquid to liquid. Often one part is water while the other can be an organic solvent. As a brief reminder the partitioning behaviour of an analyte between two phases will be reflected in LogP values as follows.
In liquid-liquid extraction the component you want to transfer called the solute here and called B in the previous explanation sits in a liquid A. It is a very big topic within analytical chemistry. Liquid-liquid extraction theory and laboratory experiments by Alders Lucas 1915-Publication date 1955 Topics Extraction Chemistry Liquids Extraction Chemistry Liquids Flüssig-Flüssig-Extraktion Extraction chimie Liquides Publisher Amsterdam Houston Elsevier Pub.
Watch this two-part series of videos from LearnChemE that shows how to use the Hunter Nash method to find the number of equilibrium stages required for a liquid-liquid extraction process. Hunter Nash Method 1. The behavioural pattern of two immiscible solvents say a and b is essentially nonideal with respect to one another.
Whats very important in liquid-liquid extraction is that the two liquids do not dissolve in one another. Liquid-liquid extraction is based on the transfer of a solute substance from one liquid phase into another liquid phase according to the solubilityExtraction becomes a very useful tool if you choose a suitable extraction solventYou can use extraction to separate a substance selectively from a mixture or to remove unwanted impurities from a solutionIn the practical. In its simplest form it may take the guise of a single stage mixing and separation unit analogous to a single stage flash in distillation.
It must have at least two components. Improvement of Density Function Theory Modeling with Experimental Validations ACS Omega. Now if a third substance is made to dissolve in a two-phase mixture of the solvents ie a and b it may behave ideally in either phases provided its concentration in each individual phase is approximately small.
The aim of this study was to provide an updated overview of the recent trends and development in LLE techniques over the past 5 years. Another common term for Liquid-Liquid extraction is as solvent extraction process. Liquid liquid extraction involves solute and solvent that are mixing together in order to separates the products.
Different variations of this. It also has applications in the isolation of natural products as in the extraction of caffeine from tea leaves. Liquidliquid extraction is possible in non-aqueous systems.
Liquid - liquid extraction is based on the transfer of a solute substance from one liquid phase into another liquid phase according to the solubilityExtraction becomes a very useful tool if. We investigate here the molecular mechanism for liquid-liquid extraction and revisit the classical two-film theory. The process involves taking liquids mixing them and being able to separate them when the liquid settles.
Liquidliquid extraction LLE is based on the principle that a solute or an analyte can distribute itself in a certain ratio between two immiscible solvents usually water aqueous phase and organic solvent organic phase. The organic product will be soluble in an organic solvent organic layer while the inorganic substances will be soluble in water aqueous layer. Liquid-Liquid extraction is a more complex process of separating a liquid mixture over the Liquid-Solid process.
Frequently one of the solvents is water or an aqueous mixture and the other is a nonpolar organic liquid. Liquid-Liquid Extraction LLX is a mass transfer operation which is used to separate components of a liquid mixture by contacting it with another liquid. Liquid-Liquid Extraction of Furfural from Water by Hydrophobic Deep Eutectic Solvents.
4 5 Extraction Theory Chemistry Libretexts

Extraction Theory Org I Lab W J Kelly Liquid Liquid Extraction Is A Useful Method To Separate Components Compounds Of A Mixture Ppt Download

Chapter 5 Liquid Liquid Extraction Ppt Video Online Download

Liquid Liquid Extraction Vs Solid Phase Extraction
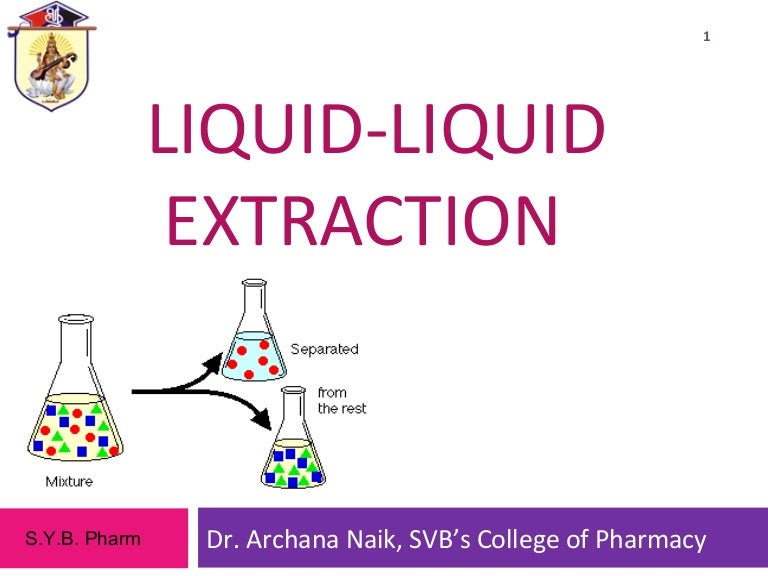
Liquid Liquid Extraction Sy 2014

Liquid Liquid Extraction Vs Solid Phase Extraction
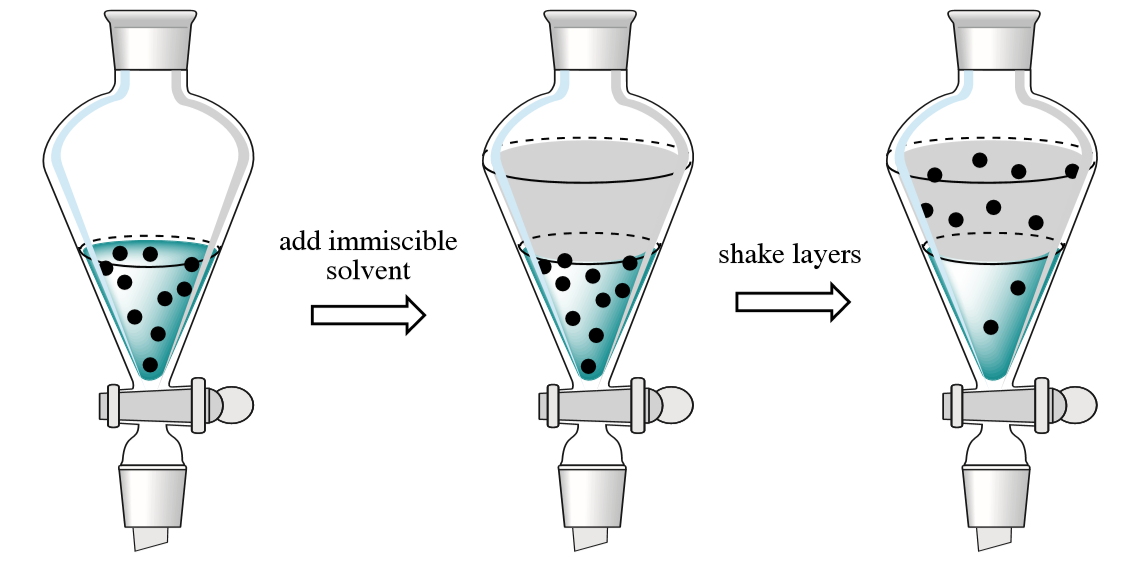
4 2 Overview Of Extraction Chemistry Libretexts

Liquid Liquid Extraction Chemical Engineering World

Liquid Liquid Extraction Sy 2014

What Is Extraction The Theory And How It S Applied To Food Foodcrumbles

Liquid Liquid Extraction Sy 2014
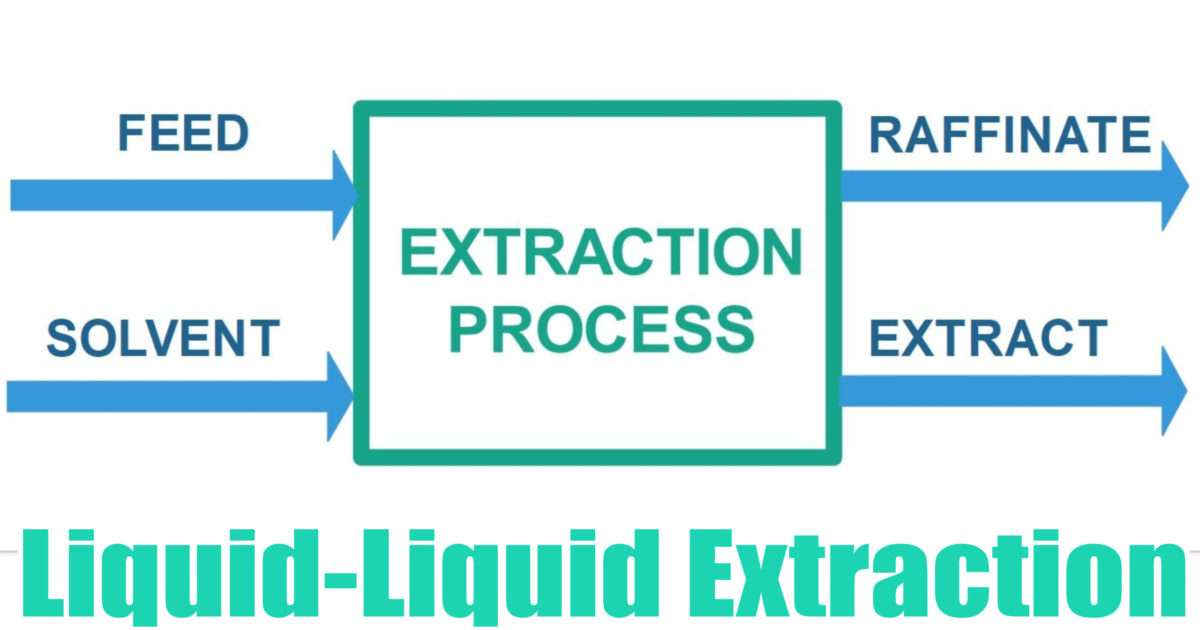
Liquid Liquid Extraction Chemical Engineering World

Liquid Liquid Extraction Vs Solid Phase Extraction

Liquid Liquid Extraction Protocol For The Removal Of Aldehydes And Highly Reactive Ketones From Mixtures Organic Process Research Development

Liquid Liquid Vs Supported Liquid Vs Solid Phase Extraction